Characteristics of slush hydrogen
Since liquid hydrogen’s density and latent heat of vaporization are approximately
1/14 and 1/5 of those of water, respectively, fuel storage tank capacities
become greater for rockets and fuel cells. Also, due to boil-off resulting
from heat inleak during transport and storage, the reduction in transport
and storage efficiency is a practical problem. For example, the densification
of liquid hydrogen could make it possible to reduce the structural weight
of rocket propellant tanks, thus allowing for an increase in payload (satellite)
weight.
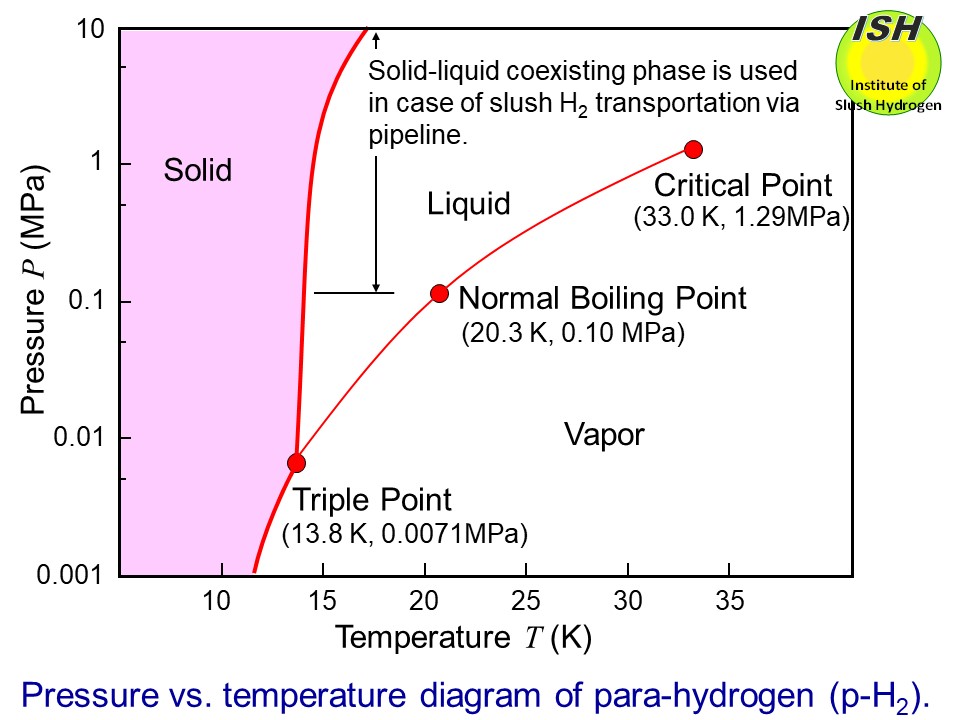
Slush hydrogen is a cryogenic solid-liquid two-phase fluid, wherein solid
hydrogen particles (particle size of about 1 mm) are contained in liquid
hydrogen, featuring greater density, and refrigerant heat capacity than
liquid hydrogen.
Compared to normal boiling liquid hydrogen (temperature of 20 K), slush hydrogen with a mass solid fraction of 50 wt.% (temperature of 14
K) features a 15% greater density, and an 18% increase in refrigerant heat
capacity (enthalpy), as shown in the above figure.
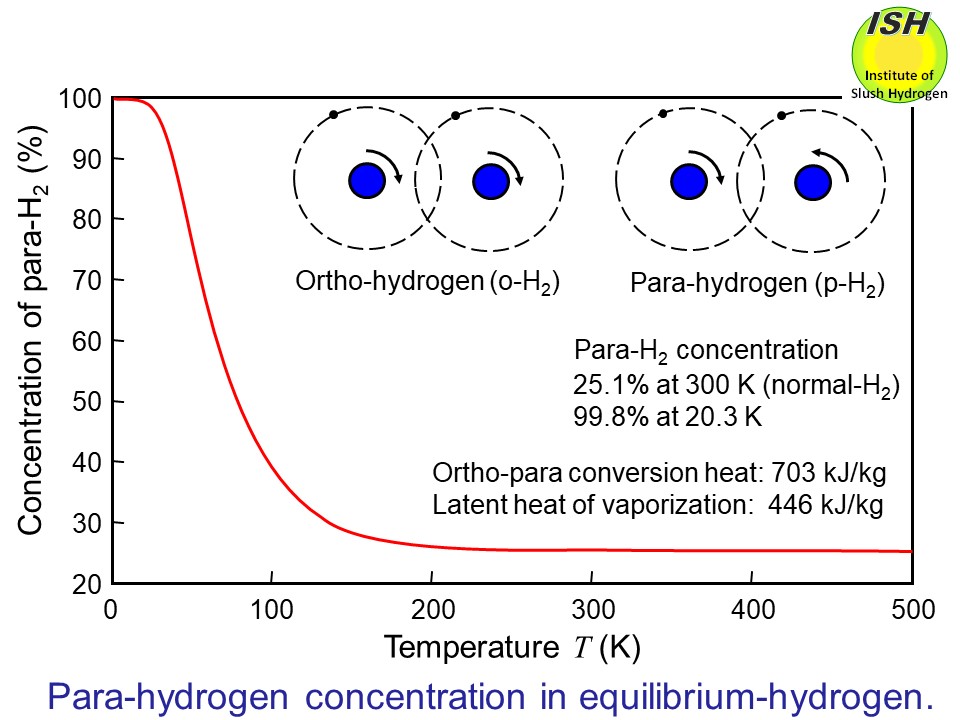
There are two different molecular forms of hydrogen: ortho-hydrogen (o-H2) and para-hydrogen (p-H2) as shown in the figure below. In the case of ortho-hydrogen, the two protons of the hydrogen molecule
spin in the same direction, while in the case of para-hydrogen, they spin
in opposite directions.
The equilibrium concentrations of ortho- and para-hydrogen differ according
to temperature. That of para-hydrogen is approximately 25% by volume at
room temperature, but nearly 100% at liquid temperature. The changeover
of ortho- to para-hydrogen generates heat of conversion of 703 kJ/kg at
the normal boiling point of hydrogen (20 K). Thus, when hydrogen gas with
a para-hydrogen concentration of 25% is liquefied and stored, ortho-para
conversion gradually occurs, generating a heat of conversion of 527 kJ/kg.
Since liquid hydrogen’s latent heat of vaporization is 446 kJ/kg, substantial
boiloff occurs during long-term storage, and storage efficiency becomes
poor.
Hydrogen liquefiers normally use a catalyst to speed up the conversion
during the liquefaction process so as to produce liquid hydrogen for storage
and transport with a para-hydrogen concentration of nearly 100%. The mixture
of ortho- and para-hydrogen at high temperatures is called normal hydrogen
(n-H2), which is a mixture of 75% ortho-hydrogen and 25% para-hydrogen, by volume.
The use of slush hydrogen would thus allow more efficient transport and
storage. Also, in the case of heat due to heat inleak or superconducting
quench*, the solid fraction would be reduced due to absorption of some
of this heat by the heat of fusion associated with the solid, but liquid
temperature increase and vapor-liquid two-phase change** would be inhibited.
Another representative slush fluid is slush nitrogen, which is being considered
for use as the refrigerant for high-temperature superconducting equipment.
In this case, also at a mass solid fraction of 50 wt.% (temperature of
63 K***), density is 16% higher than for normal boiling liquid nitrogen
(temperature of 77 K), while the refrigerant heat capacity (enthalpy) increases
by 22%.
Slush hydrogen offers superior characteristics as a functional thermal
fluid, and various applications are anticipated for hydrogen transportation
via pipeline as illustrated in the figure below and storage [4].
* Quench: Transition from a superconducting state to a normal conducting
state.
** Vapor-liquid two-phase: When the fluid is flowing in a vapor-liquid
two-phase state, larger oscillations of pressure and temperature emerge
than for those of flowing in a single-phase state, and would cause the
quench. For example, the Ledinegg-type flow instability described in the
page of "Two-phase boiling nitrogen flow and heat transfer".
*** The triple point temperature coexisting liquid, solid, and vapor of
slush nitrogen is -210℃ or 63 K. The slush nitrogen temperature coexisting
liquid and solid are, depending on the pressure, higher than -210℃ or 63
K.